
Helium is used for filling balloons and lighter-than-air craft because it does not burn, making it safer to use than hydrogen. Argon is the third most abundant gas in dry air. Fluorescent tubes commonly contain a mixture of argon and mercury vapor. ElementĪrgon is useful in the manufacture of gas-filled electric light bulbs, where its lower heat conductivity and chemical inertness made it preferable to nitrogen for inhibiting the vaporization of the tungsten filament and prolonging the life of the bulb. Table 1 summarizes the properties of the noble gases. 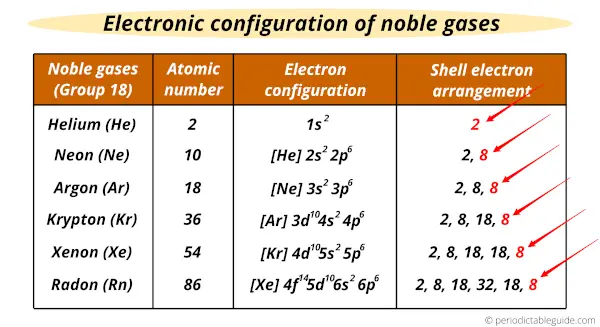
The positive electron affinity values of these elements reveal that they are unlikely to gain electrons as well. Going down the group, atomic radius increases and ionization energy decreases. These elements have the largest first ionization energies, indicating that the removal of an electron is difficult. The full s and p orbitals of the valence shell add stability to the noble gases. This is because only weak London dispersion forces are present, and these forces can hold the atoms together only when molecular motion is very slight, as it is a very low temperature. The boiling points and melting points of the noble gases are extremely low relative to those of other substances of comparable atomic or molecular masses. Its accumulation in well-insulated, tightly sealed buildings, however, constitutes a health hazard, primarily lung cancer. More recently, it was observed that this radioactive gas is present in very small amounts in soils and minerals. Radon comes from other radioactive elements. Helium is isolated from natural gas by liquefying the condensable components, leaving only helium as a gas. Some natural gas contains 1–2% helium by mass. These elements are present in the atmosphere in small amounts. Neil Bartlett at the University of British Columbia proved this assumption to be false. They earned the name “noble” because they were assumed to be nonreactive since they have filled valence shells. The elements in group 18 are noble gases (helium, neon, argon, krypton, xenon, and radon). 
For instance, xenon hexafluoride reacts with water, yielding a solution of xenon trioxide. Xenon-compounds with the electronegative element, oxygen, can be produced by replacing fluorine atoms in xenon fluorides with oxygen. Other compounds like xenon tetrafluoride and xenon hexafluoride can also be prepared similarly. Xenon difluoride, obtained by heating an excess of xenon gas with fluorine gas, is a stable, crystalline material. For example, xenon, with the lowest ionization energy from the noble gases, was found to react with the most electronegative element, fluorine. However, in the early sixties Neil Barlett discovered some exceptions. Noble gases were Initially thought to be entirely chemically unreactive and were called inert gases. Helium is used to create an inert atmosphere during the melting and welding of easily oxidizable metals. For instance, argon is used to manufacture gas-filled electric light bulbs to prevent the oxidation of tungsten filaments, prolonging the bulb’s life. The high stability of noble gases attests to their chemical inertness, which finds many industrial applications. Noble gases resist electron additions as their valence shells are already complete, and the incoming electron needs to enter a higher principal quantum shell. Meaning, energy is required to add an additional electron to a gaseous atom. Noble gases also have positive electron affinity values. The removal of an electron requires the input of a large amount of energy, which is unfavorable. This is because these elements have stable electron configurations with complete octets. Yet, noble gases have high first ionization energies compared to all other elements in the periodic table. Moving down the group, the elements exhibit an increase in boiling points, densities, and atomic radii, which consequently leads to the decline of ionization energies of each successive element. Radon is the only radioactive element from group 18. These elements occur as monatomic species and exist as gases under room-temperature. The nonmetallic elements categorized under group 18 – helium, neon, argon, krypton, xenon, and radon – are called noble gases.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
